|
The information in this guide is especially helpful for labs that use HPLC’s, GC’s, MS’s, and similar devices to test samples. Therefore, I have created a guide to help chemical laboratories estimate uncertainty in measurement for their analytical processes. 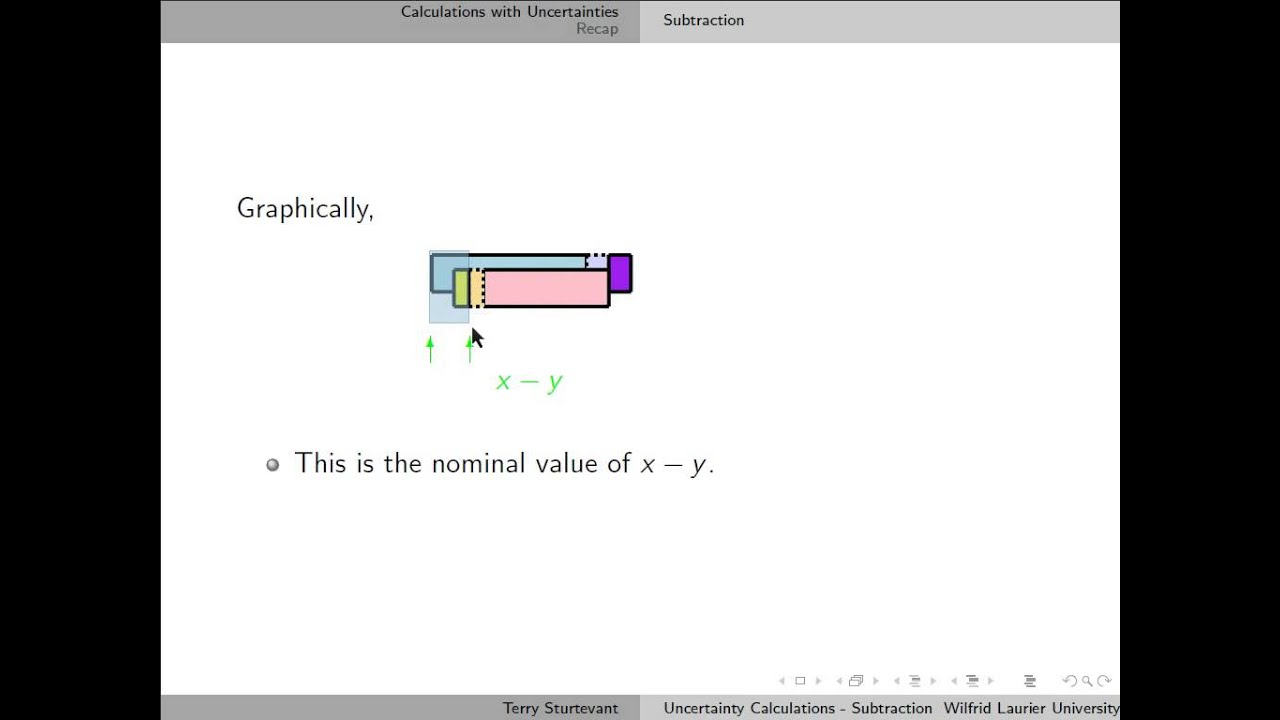
This creates a gap in the market that leaves many chemical laboratories without a solution or examples to estimate uncertainty. The problem for chemical labs is the availability of guides and resources is limited which can make estimating uncertainty difficult. There is a multi-step process that needs to be evaluated from beginning to end. However, when you estimate uncertainty for analytical measurements, the process is not same. Additionally, there are plenty of guides and resources available to help with uncertainty analysis. mass, volume, temperature, pH, etc.), the process is similar to the methods used by other types of laboratories. When you evaluate uncertainty for physical measurements (e.g. Although each uncertainty analysis uses the GUM method, the process needed to estimate uncertainty is quite different. 
Typically, there is one process to evaluate uncertainty for physical measurements and another process for analytical chemistry measurements. 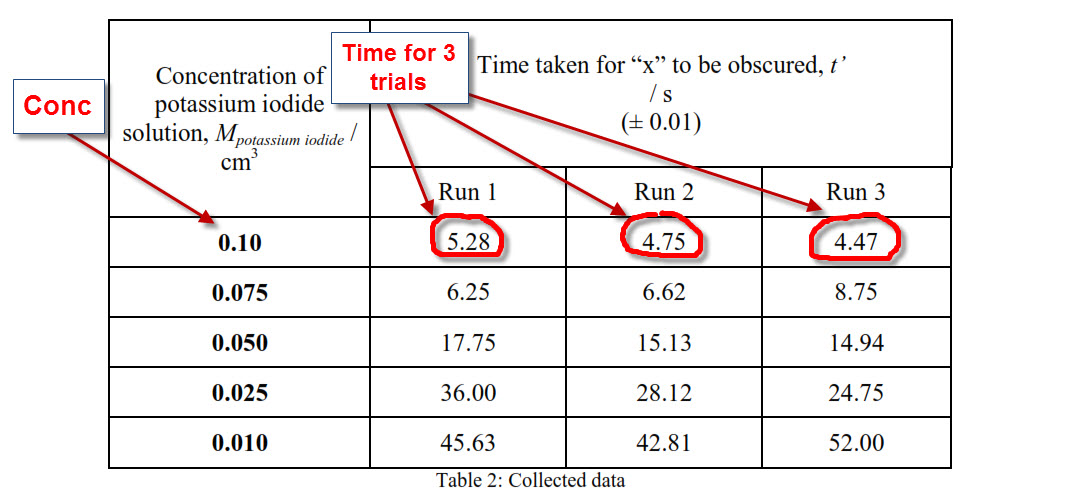
Most chemistry labs need to use two different processes to estimate uncertainty. Estimating uncertainty in measurement for chemistry laboratories is quite different compared to other types of laboratories.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
